|
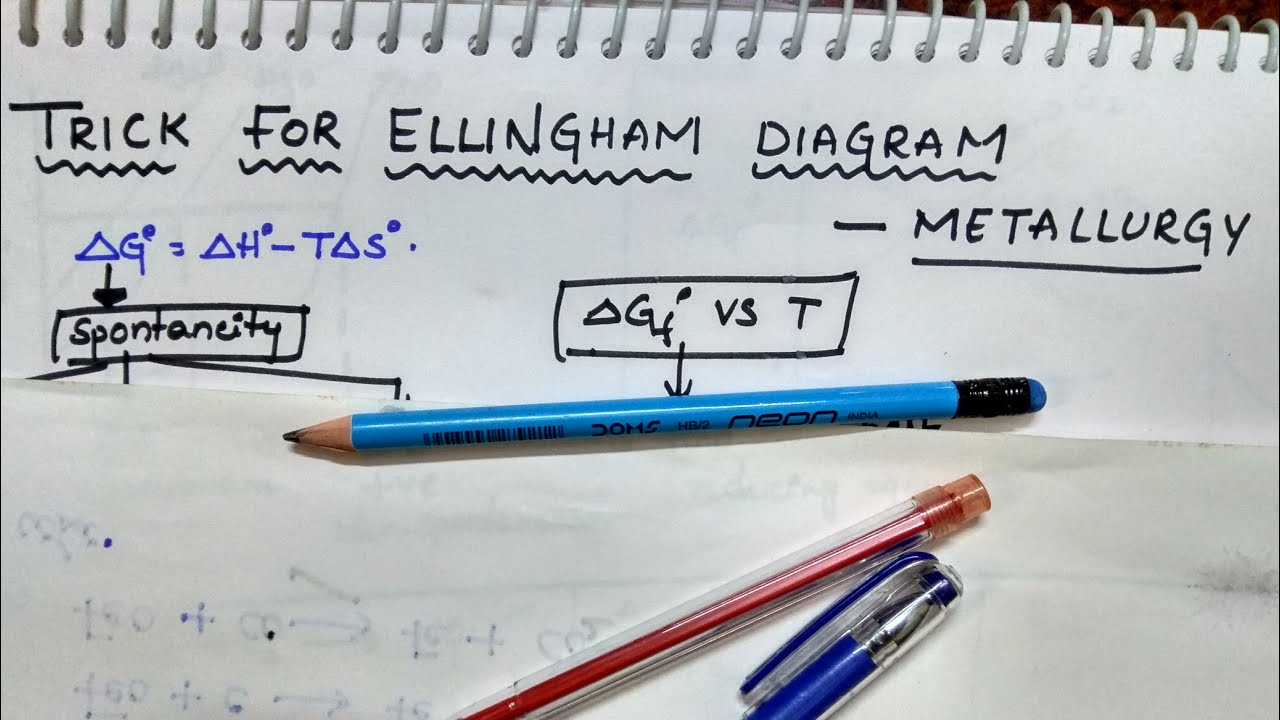
1/5/2024 0 Comments Ellingham diagram explanation The Ellingham diagram shows Gibbs free energy modification (G) because the temperature operates in every chemical reaction method. Now if ∆G°>0 for A + B \ C then this reaction will be non – spontaneous.Ĭ \ A + B, then ∆G°<0 so the reaction will be spontaneous.įor example, suppose a graph is given for following reactions of metal oxides –Įllingham diagram area unit, a picture of a thermodynamic conception. If ∆G☀ so the reaction will be non-spontaneous. General representation is shown below for your better understanding – Values above the ∆G°=0 will be positive while below the ∆G°=0 will be negative. Thus, in a way, the Ellingham diagram relates thermodynamics and metallurgy. In this Ellingham diagram plays an important role to select a best suitable reducing agent. For this purpose, metal oxides and sulfides are reduced by best suitable reducing agents through spontaneous reactions. In metallurgy, we want metal oxides and sulfides to get reduced to get pure metal at low cost. If ∆G☀, then the reaction will be non-spontaneous. It helps us to know the best reducing agent for a particular metal oxide and sulfides. analysis could be about thermodynamic surroundings and doesn't embrace reaction mechanics.Įllingham diagram is a plot between Gibbs free energy and temperature. Diagrams are accustomed to predict how a metal will regenerate into a containing metal. In metals, the Ellingham diagram is employed to calculate the equilibrium temperature between the metal, its oxide, and oxygen, also because of the interaction between iron and sulfur, nitrogen, and different non-metals. The primary of those statues was fictionalized by Harold Ellingham in 1944. This technique is usually accustomed to confirm however simple it's to cut back iron oxides and sulfides.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
